a)-(d). Production rates of • OH, O, H • and H 2 over a wide range of... | Download Scientific Diagram

SOLVED: Exercise 2 a) Convert the following pressure to Pa, torr, mmHg, and atm: 25 psi 42 psi 75 psi b) Convert the following pressure to psi and atm 250 KPa, 22.7 Pa 230 mmHg, 385 torr

SOLVED: Consider 3 mol of monoatomic ideal gas that is taken Irom stale A (Pa 2 atm. 10 L) to state B (Pr = atm: VR 30 L)as shown below Pa =

Bonita's Services - ATM: 👉PA-Grocery ng 1-2 weeks essential goods 👉PA-Bili ng gamot and maintenance ng aming mahal na Senior ciitizens Tips: 📝Lists in a plain paper all your grocery list 🥦Dessiminate

Gases Pressure (5.1) Gas Laws of Boyle, Charles, Avogadro (5.2) Ideal Gas Law (5.3) Gas Stoichiometry (5.4) Kinetic Molecular Theory (5.6) Effusion & Diffusion. - ppt download
for a gaseous phase reaction at 27 TEMP 2A gives 3B if pressure of A changes from 2atm to 1.5 atm in 10 sec cal. a. rate of reaction in 1. M/sec
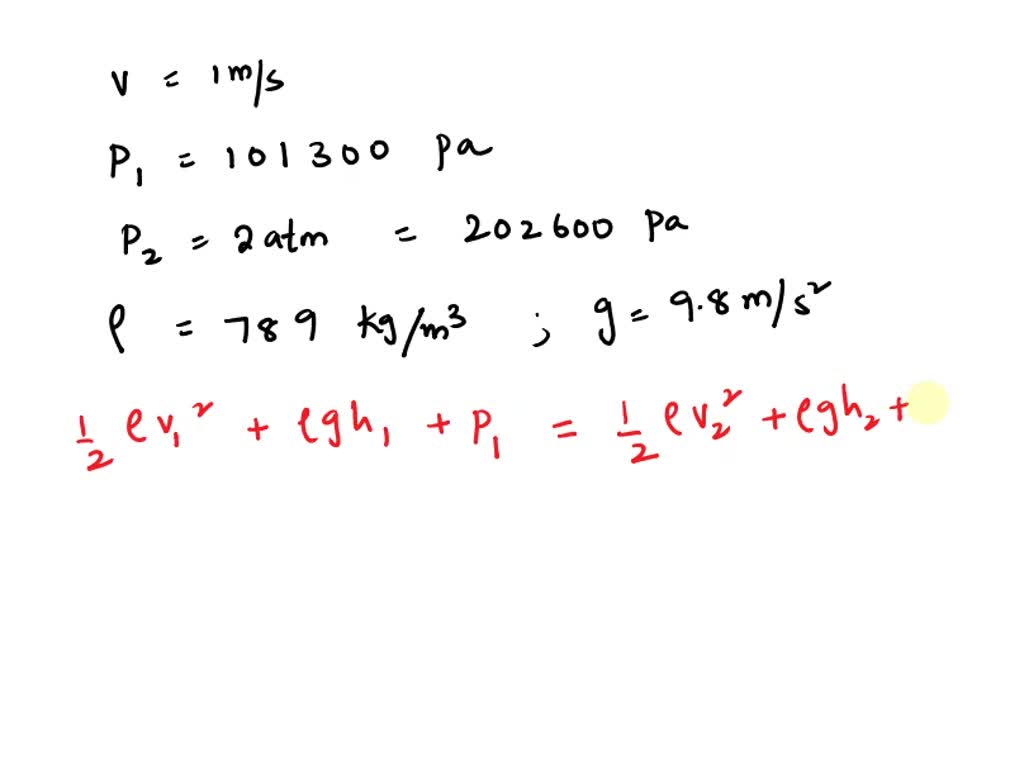










:max_bytes(150000):strip_icc()/sun-beams-diffuse-ocean-micronesia-palau-128941208-587b91ad5f9b584db36312c9.jpg)


:max_bytes(150000):strip_icc()/GettyImages-124564231-58b5c7d75f9b586046caddb9.jpg)