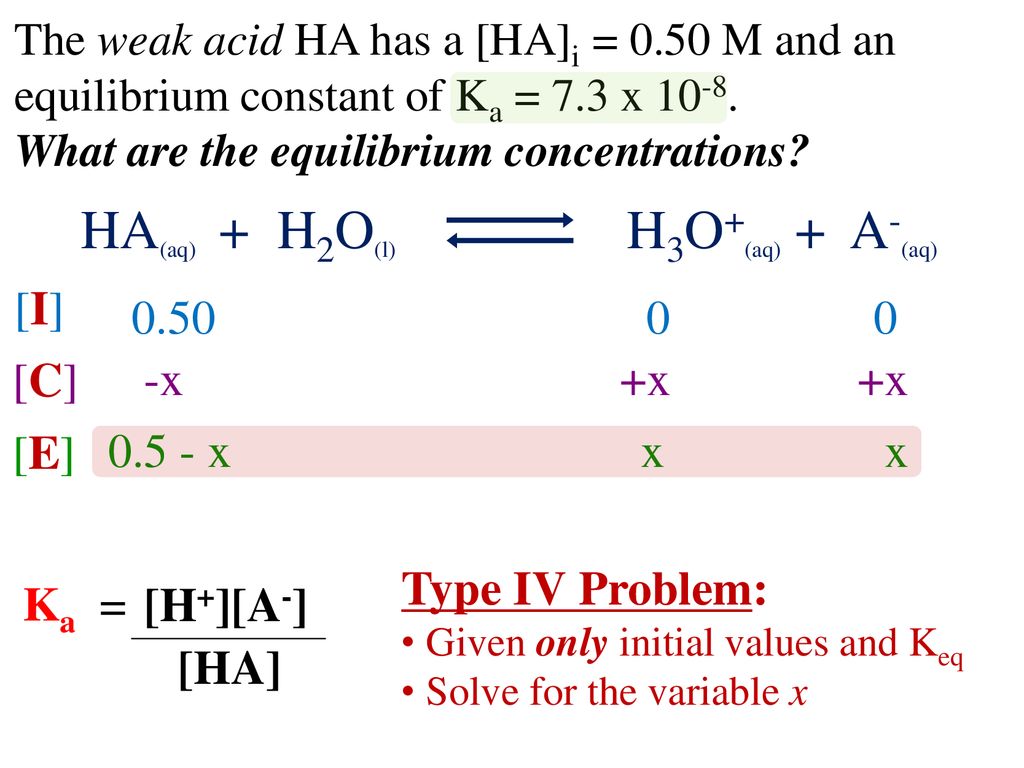
![SOLVED: For the dissociation reaction of a weak acid in water; HA(aq) + H2O(1) = Hs0+(aq) +A: (aq) the equilibrium constant is the acid-dissociation constant; Ka and takes the form [Hzo+J[A-] [HAJ SOLVED: For the dissociation reaction of a weak acid in water; HA(aq) + H2O(1) = Hs0+(aq) +A: (aq) the equilibrium constant is the acid-dissociation constant; Ka and takes the form [Hzo+J[A-] [HAJ](https://cdn.numerade.com/ask_images/b6e72b4272b24ff5a54297778f90c0b4.jpg)
SOLVED: For the dissociation reaction of a weak acid in water; HA(aq) + H2O(1) = Hs0+(aq) +A: (aq) the equilibrium constant is the acid-dissociation constant; Ka and takes the form [Hzo+J[A-] [HAJ

An adiabatic container fitted with a movable adiabatic piston (operating at 1 atm ) is filled with 2 litre of 2 M H2O2 (aq) solution at 300 K. If H2O2 dissociates following

Additional Qualifications – Answers to Some Frequently Asked Questions - OISE Continuing and Professional Learning













![simple a, aq, aaa, aaq nitials geometric network - Stock Illustration [61606783] - PIXTA simple a, aq, aaa, aaq nitials geometric network - Stock Illustration [61606783] - PIXTA](https://en.pimg.jp/061/606/783/1/61606783.jpg)