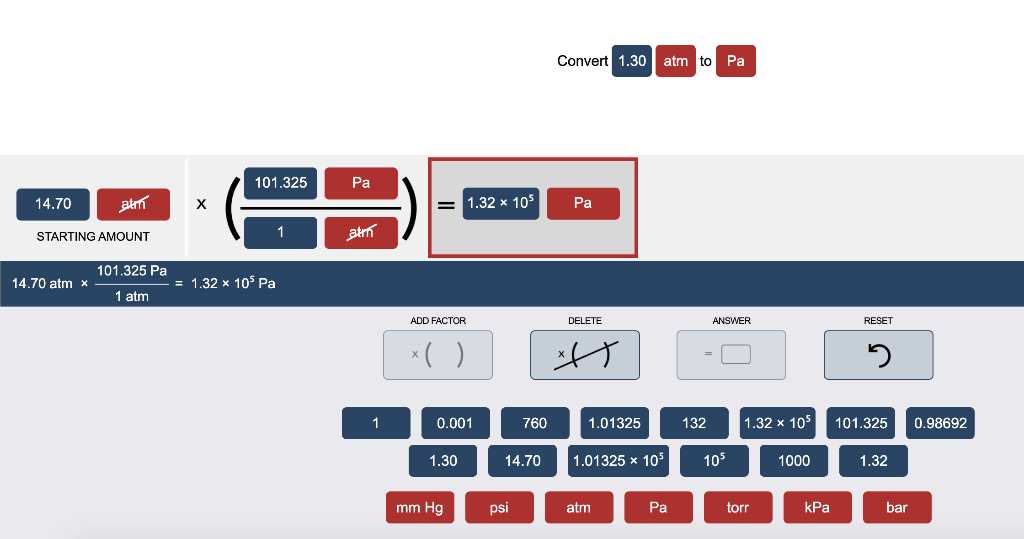
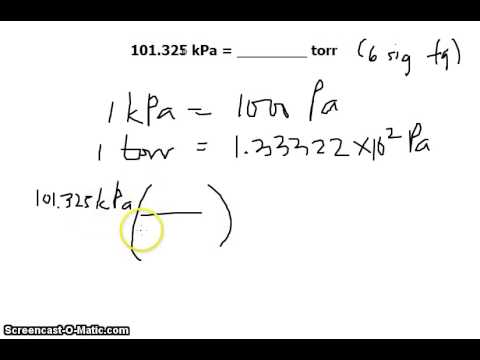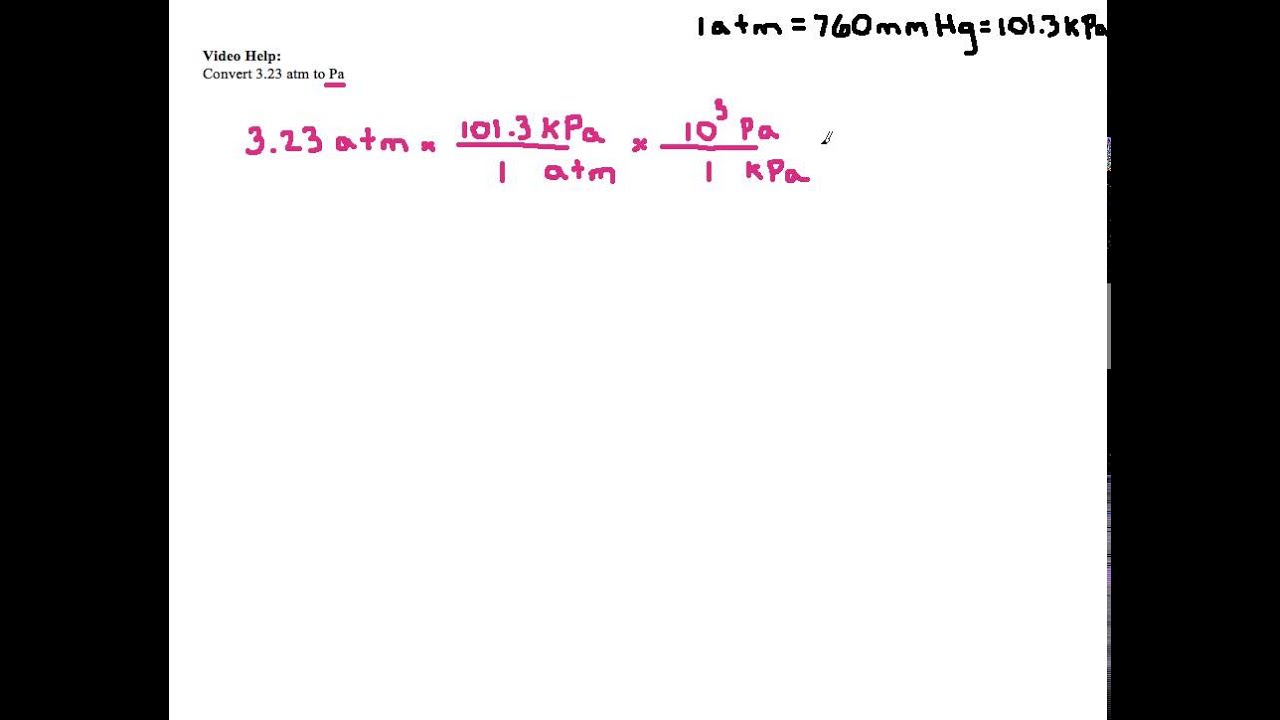SOLVED:Convert the following pressures into units of kilopascals. a. 2.07 ×10^6 Pa c. 10.9 atm b. 795 mm Hg d. 659 torr

Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download
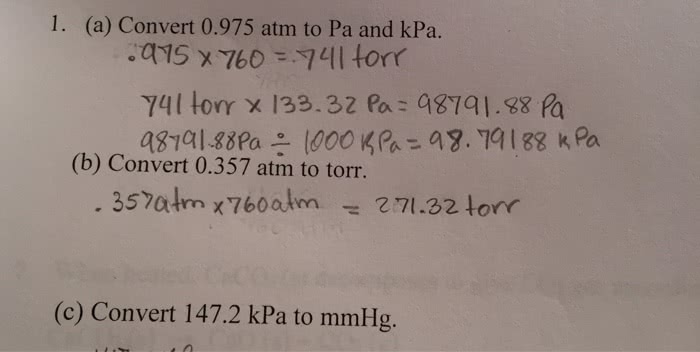
OneClass: 1. (a) Convert 0.975 atm to Pa and kPa. .qy5 x 760 = 41 torr (b) Convert 0.357 atm to torr....

SOLVED: quESt kPa? (1 atm 1.01325 measured be 654 mmHg What I5 the pressure The pressure of a gas sample Was 87.2 kPa 6,63 * 10*KPa 8.72 + 107 kPa 8,72 * 104KPa 118 KPa

SOLVED: Exercise 2 a) Convert the following pressure to Pa, torr, mmHg, and atm: 25 psi 42 psi 75 psi b) Convert the following pressure to psi and atm 250 KPa, 22.7 Pa 230 mmHg, 385 torr