
A steel wire of length 5m is pulled to have an extension of 1mm. Its Y is 1.9 x 10 ^4 N/m ^2 . The energy per unit volume stored in it is

Profiles of eigen value, pressure gradient magnitude (Pa/m) and heat... | Download Scientific Diagram

Official and provisional methods of analysis. § &. -^ e a£ ss -gg □e i is §§□* 1 S* ..s s§ □s m © ?c« a . 5 . v.o M« *

Temperature and thermal expansion Specific Heat Capacity Phase changes and Heat Molecular picture of a gas Ideal gas law Kinetic theory of. - ppt download

Video of the Week - Dr. Nicholas J. Shaheen on Premalignant Lesions of the Esophagus - American College of Gastroenterology
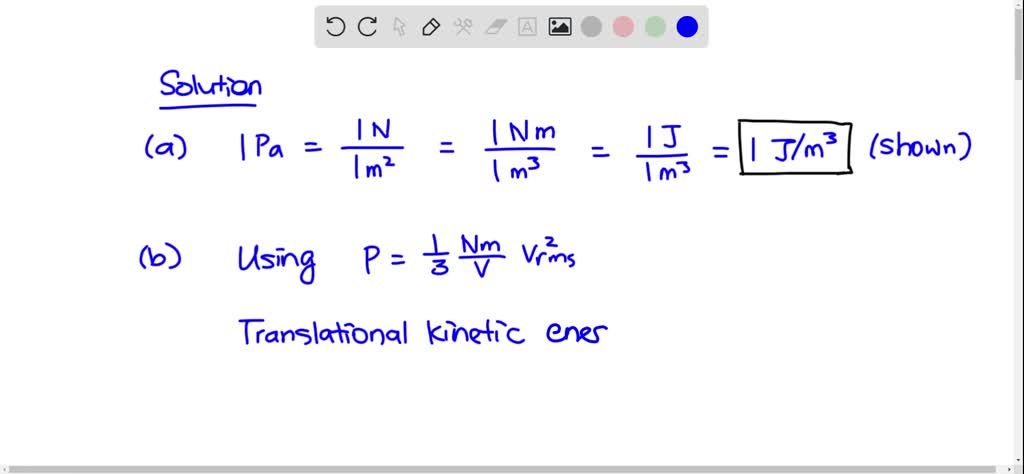
SOLVED:(a) Show that 1 Pa 1 J/m3. (b) Show that the density in space of the translational kinetic energy of an ideal gas is 3P/2.

Basics of Vacuum Technology Unit: Pa = N/m 2 = J/m 3 UnitPa or N/m 2 bar10 5 mbar100 atm= 760 torr x ppt download

A thermodynamic process is shown in the given figure. The pressures and volume corresponding to some points in the figure are:P A =3 × 104 Pa , P B =8 × 104





![ANSWERED] A chemical reaction transfers 7850 J of thermal ener... - Physics ANSWERED] A chemical reaction transfers 7850 J of thermal ener... - Physics](https://media.kunduz.com/media/sug-question/raw/52881699-1659022219.4228623.jpeg)






