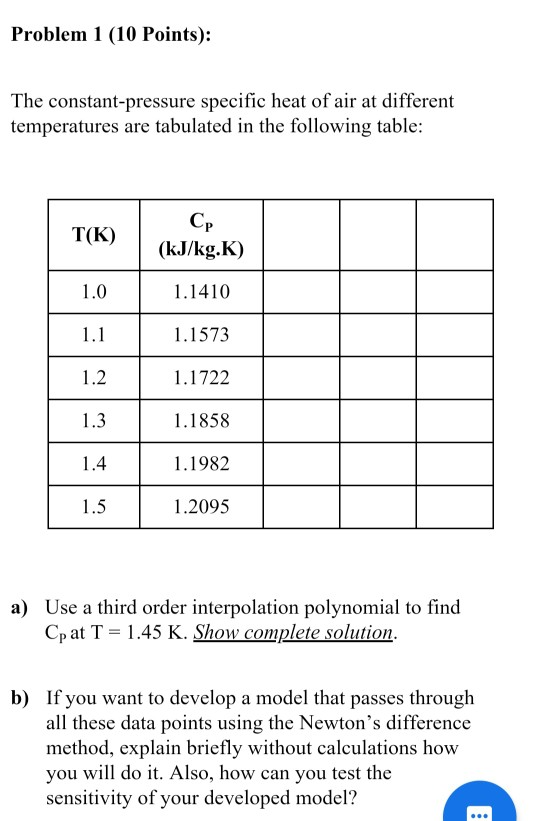
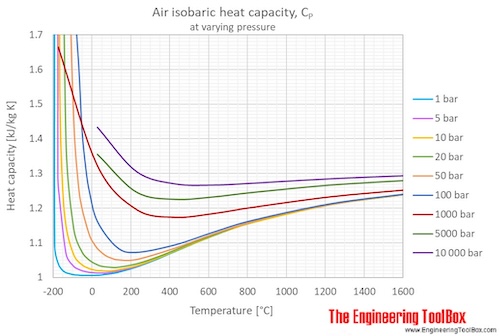
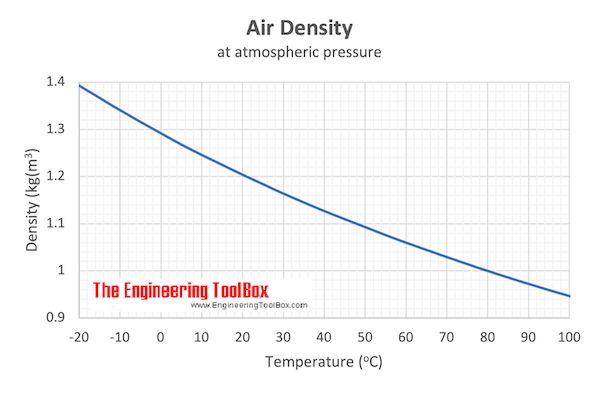
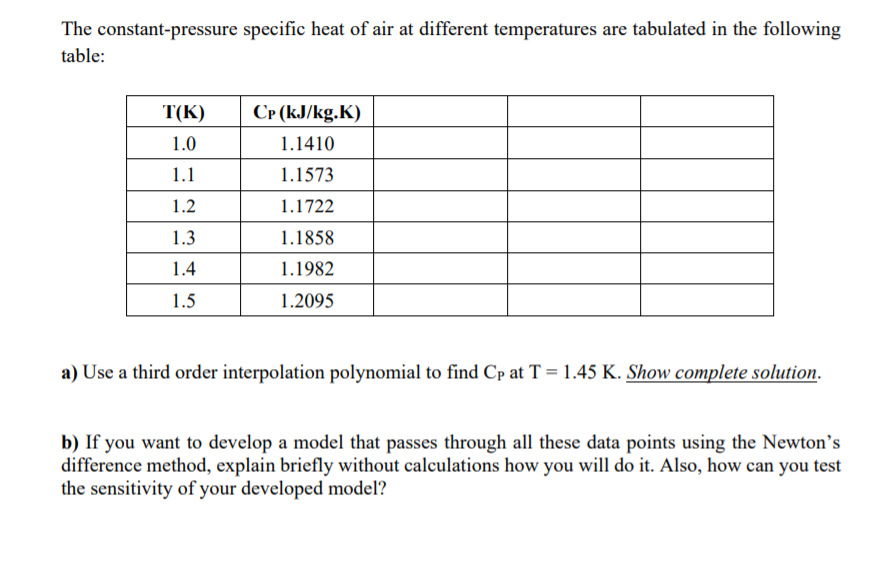
If the specific heat capacity of air at constant pressure is 993 J kg ^(-1) K ^(-1) calculate specific heat capacity at constant volume ? Density of air at N.T.P. is 1.293 Kg //m ^(3). [E.Q.)

1: Specific heat at constant pressure vs. temperature for air and CO 2 | Download Scientific Diagram

The specific heat at constant pressure and at constant volume for an ideal gas are C_(p) and C_(... - YouTube

Estimation of air specific heat ratio at elevated pressures using simple predictive tool - ScienceDirect
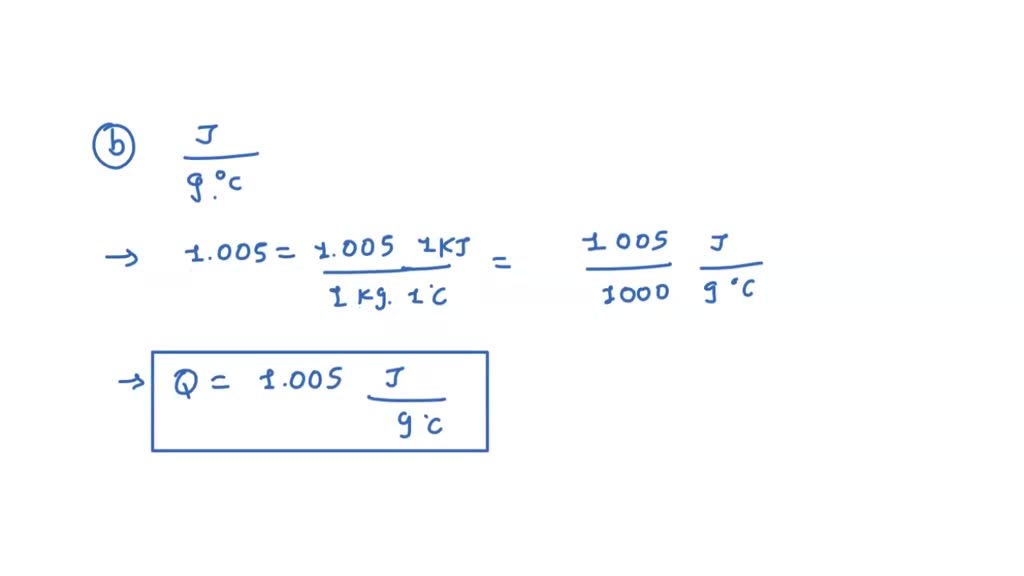
SOLVED: The constant-pressure specific heat of air at 25^∘C is 1.005 kJ / kg·^∘C . Express this value in kJ / kg·K, J / g·^∘C, kcal / kg·^∘C, and Btu / lbm·^∘F.

The specific heat of air at constant pressure is `1.005 kJ//kg//K` and the specific heat of air at - YouTube

Variation in specific heat at constant pressure with temperature for... | Download Scientific Diagram

SPECIFIC HEATS The specific heat is defined as the energy required to raise the temperature of a unit mass of a substance by one degree. In general, this. - ppt download

From given data, calculate the value of the mechanical equivalent of heat. The specific heat capacity of air at constant volume is 170 cal/kg - K, gamma = CP/CV = 1.4 and