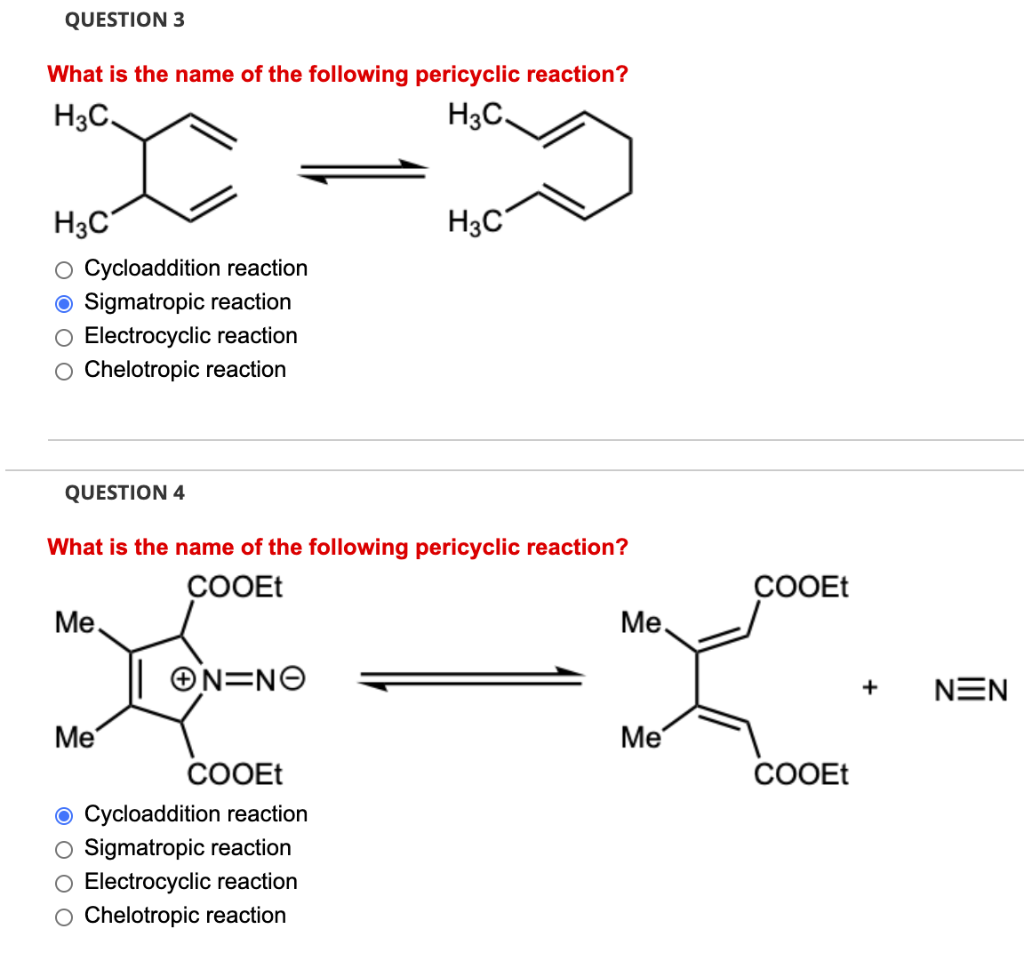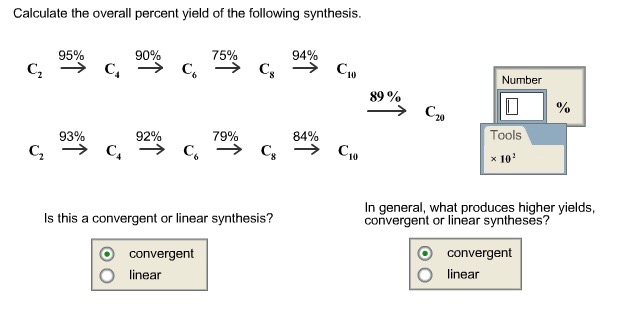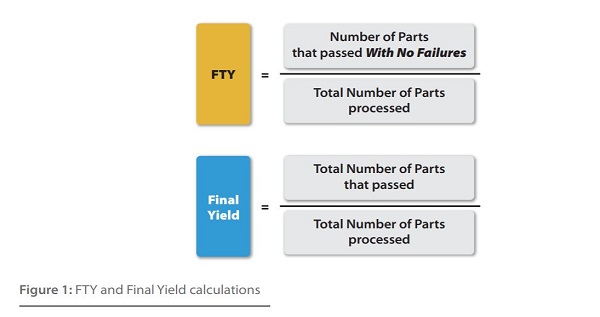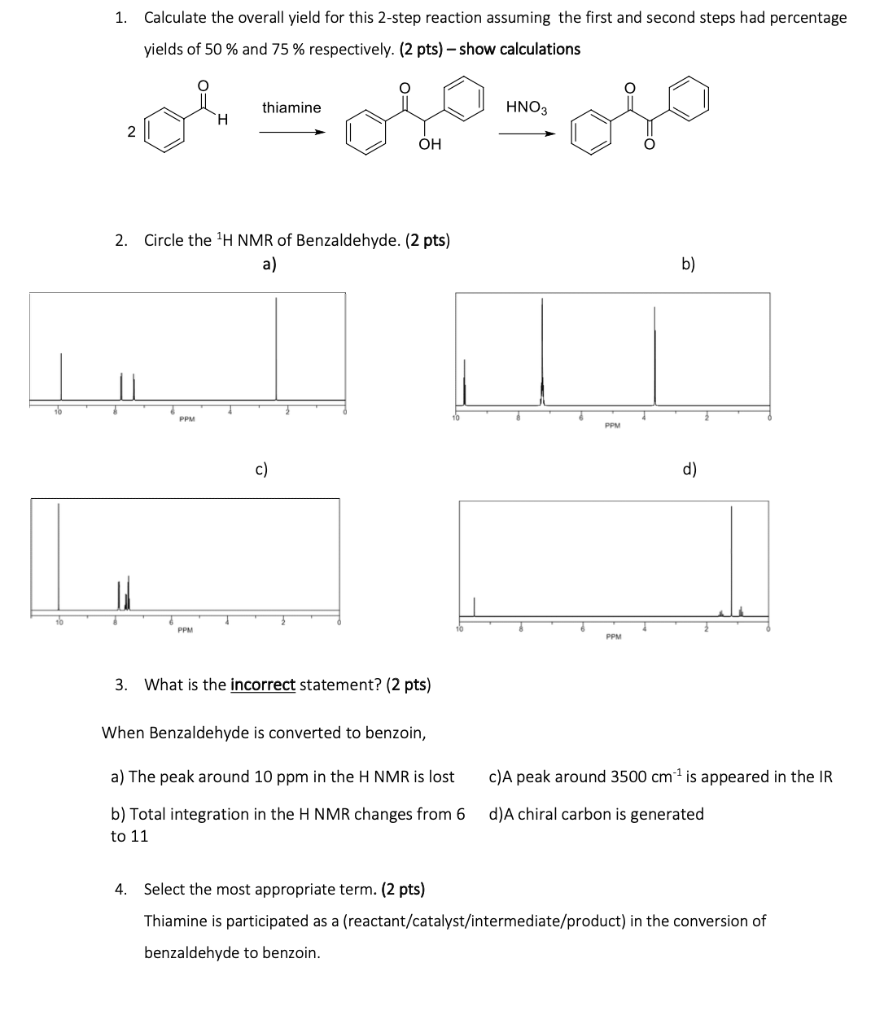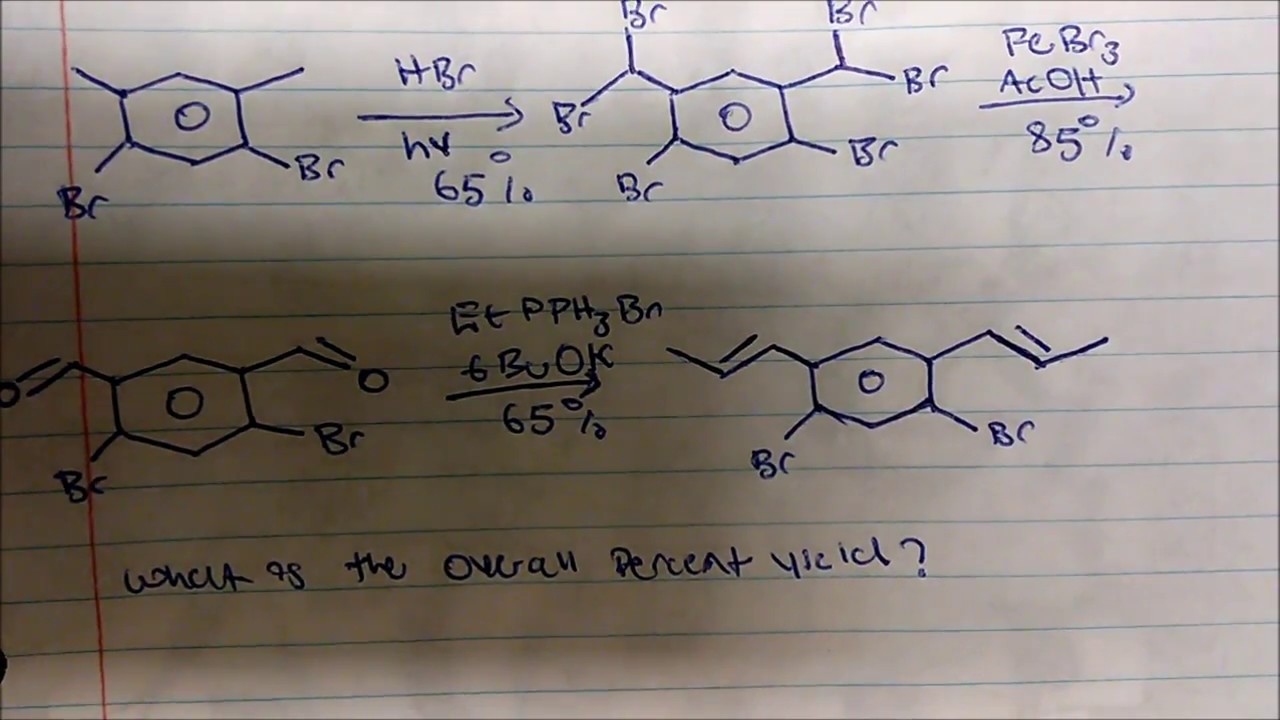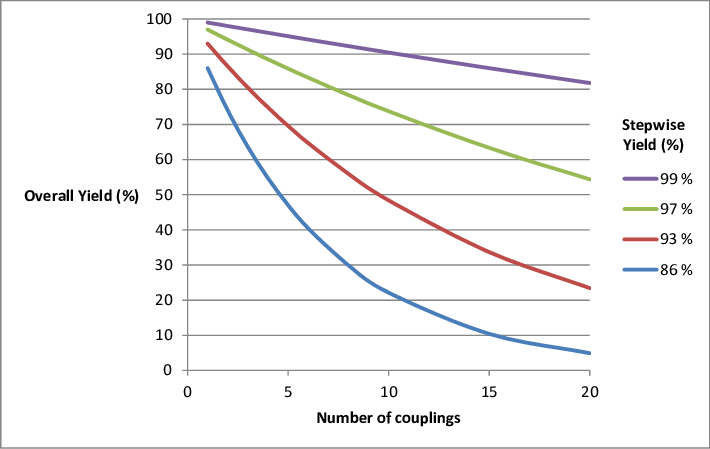
Overall yield vs number of steps coupling yield at different stepwise... | Download Scientific Diagram

For a sequential reaction : A rightarrow B + C 2B rightarrow C + 2D If % yeild of (i) and (ii) reactions are 90% and 80% respectively, then the overall % yield is expected to be:

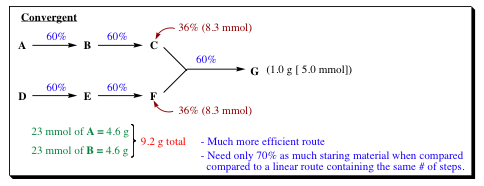
SOLVED: The overall percent yield for a multistep synthesis can be calculated by multiplying the decimal equivalent of percent yield for each step together and then multiplying by 100 to get the
![A-level Chemistry] Step and Overall Yield- I don't understand the answer to part B. 0.65/0.75 x100, but why does that work? : r/HomeworkHelp A-level Chemistry] Step and Overall Yield- I don't understand the answer to part B. 0.65/0.75 x100, but why does that work? : r/HomeworkHelp](https://preview.redd.it/a-level-chemistry-step-and-overall-yield-i-dont-understand-v0-jc4ld8u88ixa1.jpg?auto=webp&s=838996dfc2238aa519e108cb734d1614f8b3d24c)
A-level Chemistry] Step and Overall Yield- I don't understand the answer to part B. 0.65/0.75 x100, but why does that work? : r/HomeworkHelp
In the following reaction: The % yield for reaction I is 60% and that of reaction II is 50%. The overall yield of the complete reaction is ___% - Sarthaks eConnect | Largest Online Education Community
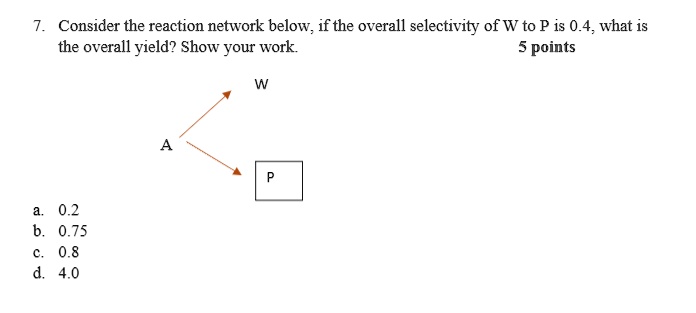
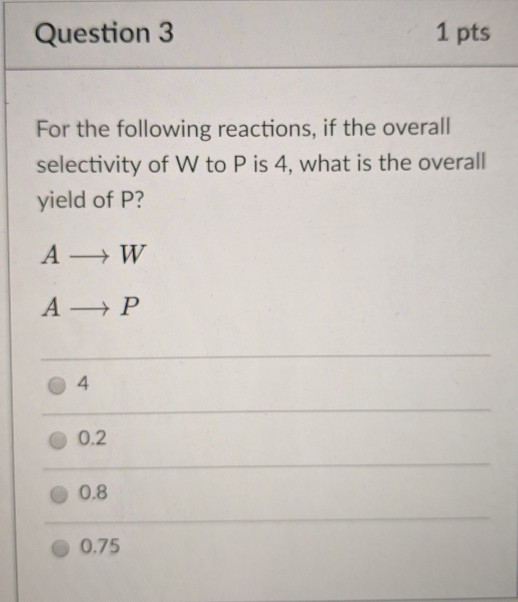
SOLVED: I. Consider the reaction network below,if the overall selectivity of W to P is 0.4,what is the overall yield? Show your work 5 points W A a. 0.2 b. 0.75 c. 0.8 d. 4.0