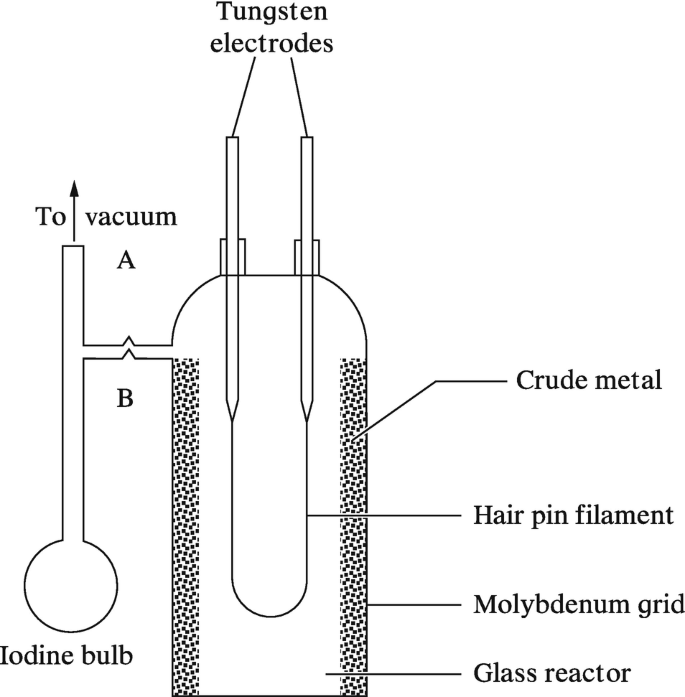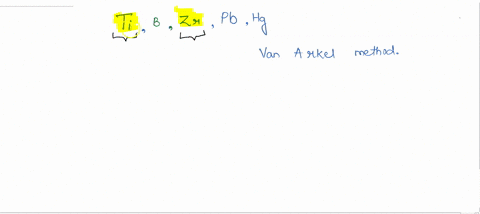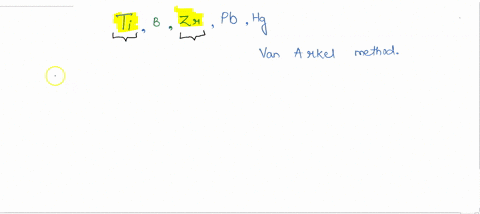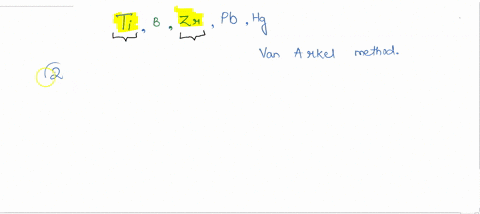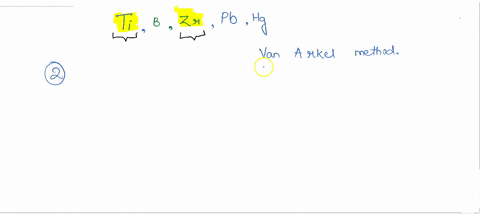
SOLVED:How many metals are commercially purified by Van Arkel method from the given metals? Ti, B, Zr, Pb, Hg
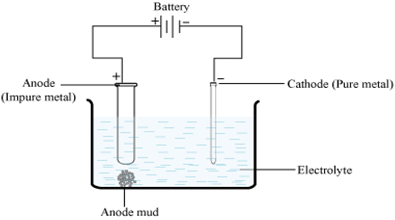
What is electro-refining? Explain with one example. from Chemistry General Principles And Processes of Isolation Of Elements Class 12 Jammu and Kashmir Board

SOLVED: Among the metals, Ti, V, W, Zr, Th and Au, the no. of metals purified by van Arkel method is

In van Arkel method, if `I_(2)` is introducted at `1800K` anode impure zironium metal, the product - YouTube

Respected Ma'am what is zone refining and van arkel method Actually I didn't understand it - Chemistry - - 15658505 | Meritnation.com
%2BMond%2BProcess%2Band%2B(2)%2B%2Bvan%2Barkel%2Bmethod.png)
Chemistry Notes Info - Your Chemistry Tutor provide notes for 9,10,11,12, BSc, MSc, Chemistry Quiz: 12 Class Chapter 6- General principle and process of isolation of the elements