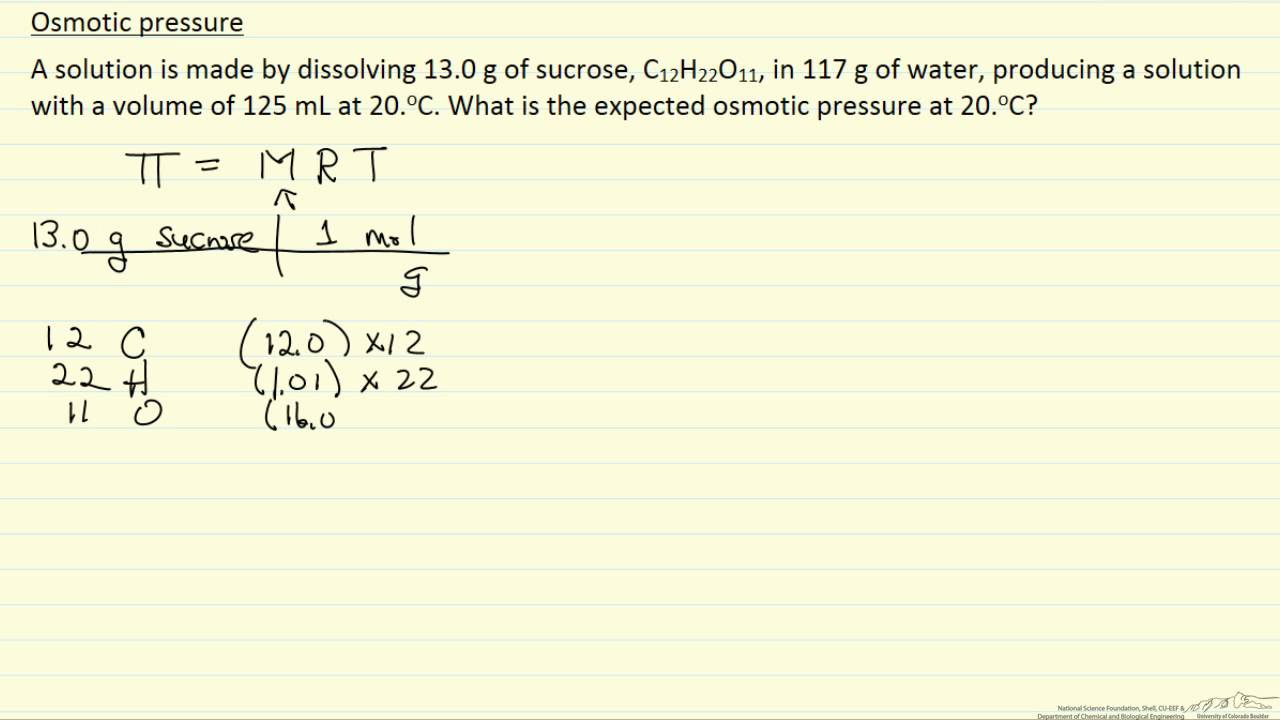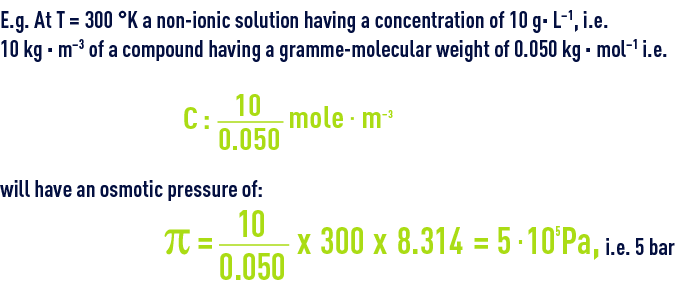SOLVED: Membrane Separation II Reverse Osmosis (a) Derive van Hoff' s equation (osmotic pressure T = cRT) (b) feed solution containing Sg NaCI/L (p-1OOlkg/m ) is purified using reverse osmosis system: Water

SOLVED: TC = g C o RT van't Hoff equation Osmotic pressure (atm) of particles per mole solution (Osm mol) Concentration(mmol Reflection coefficient (vares from Gas constant 082 atm mol Absolute temperature (

van't Hoff proved that osmotic pressure (pi) is a colligative property. For an ideal solution, osmotic pressure(pi) is helpful to determine that molecular mass of solute using M(B)=(W(B)RT)/(pi.V) Relation can exxpressed by