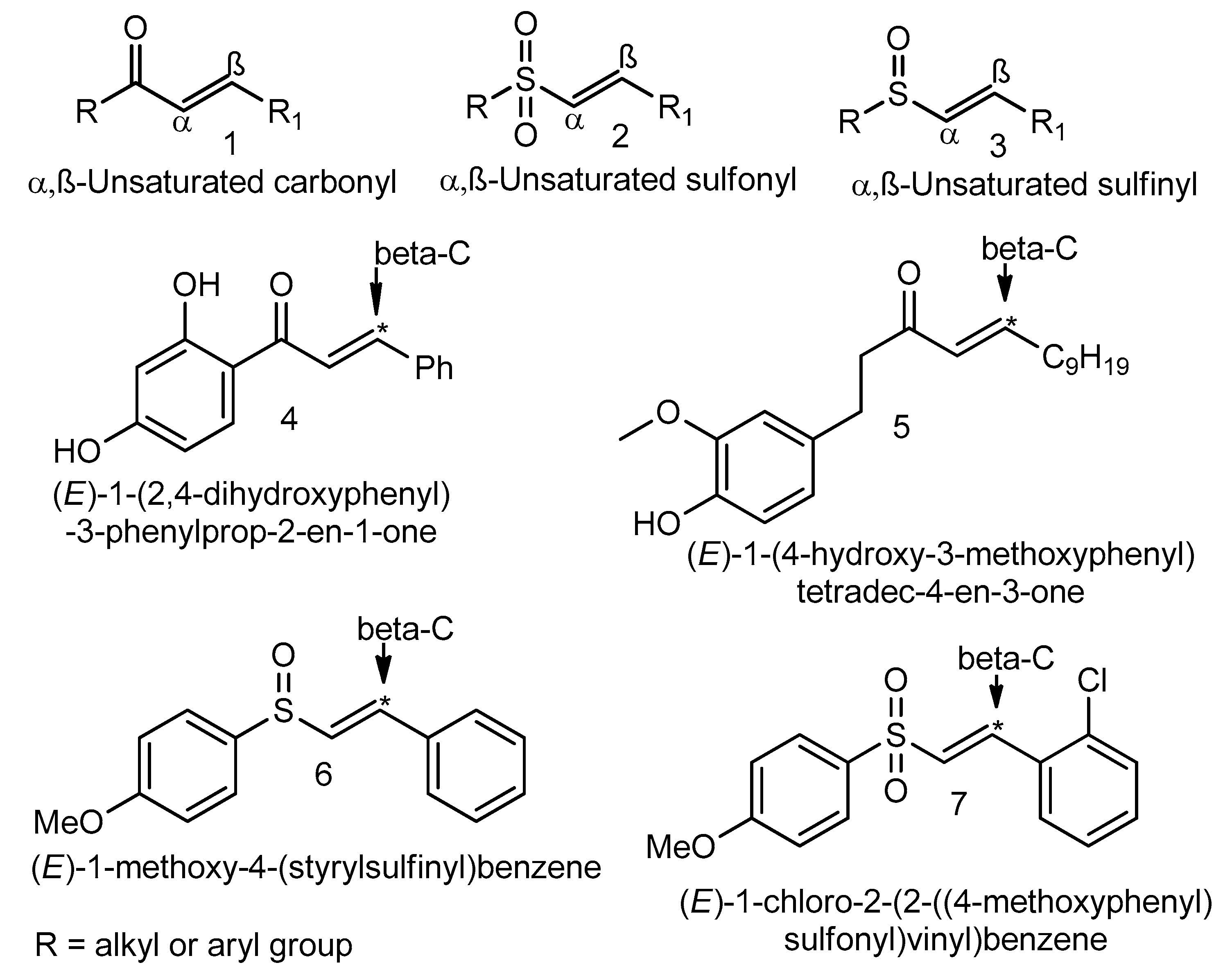
Synthesis of alkenes: claisen rearrangement of allyl vinyl ethers, part i; mechanistic views; the organic chemistry notebook series, a didactical approach, N2 9
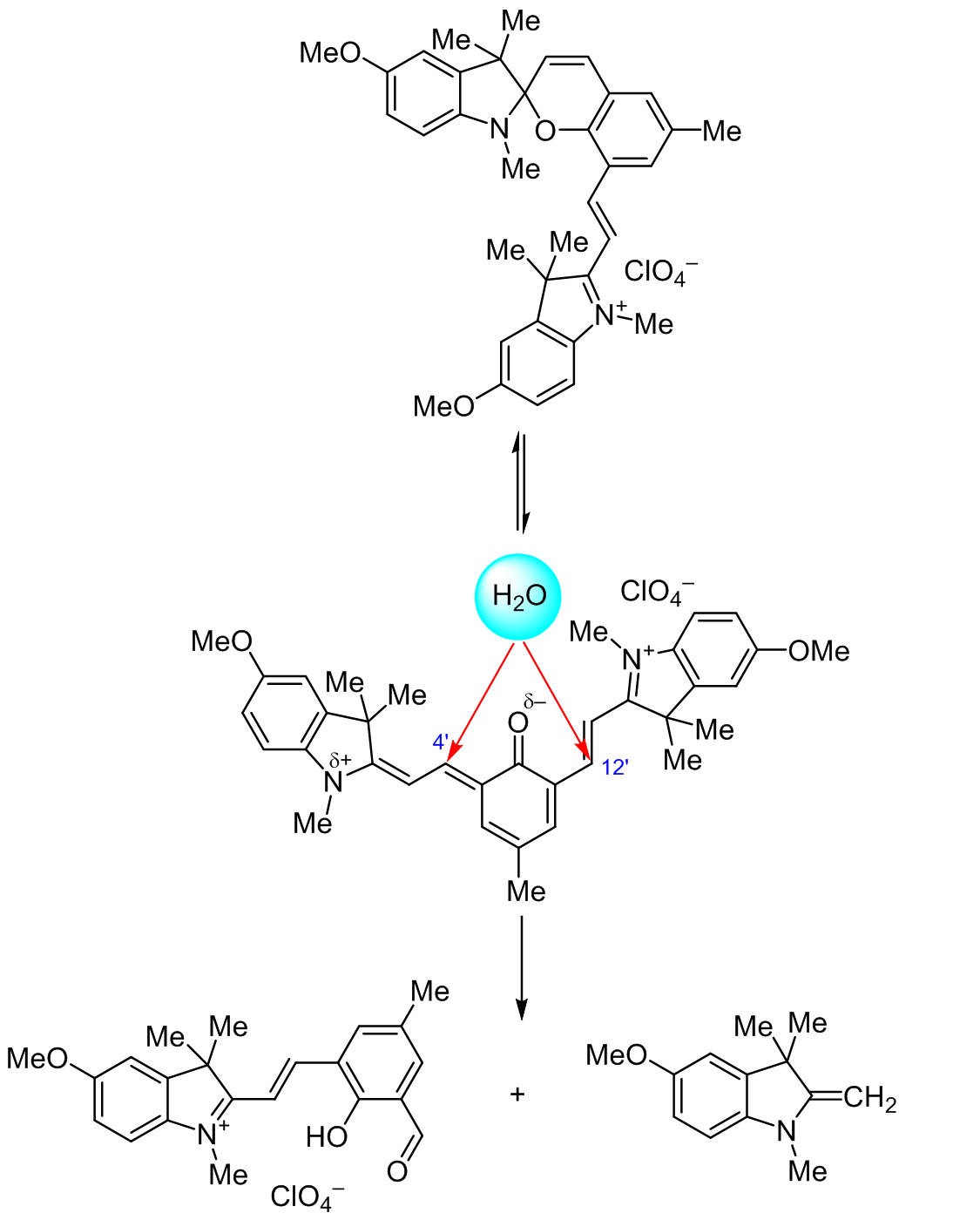
SYNTHESIS AND STRUCTURAL CHARACTERIZATION OF NEW SPIROPYRAN CONTAINING CONJUGATED VINYL-3<i>Н</i>-INDOLIUM MOIETY AND ITS HYDROLYSIS PRODUCT | Козленко | Chemistry of Heterocyclic Compounds
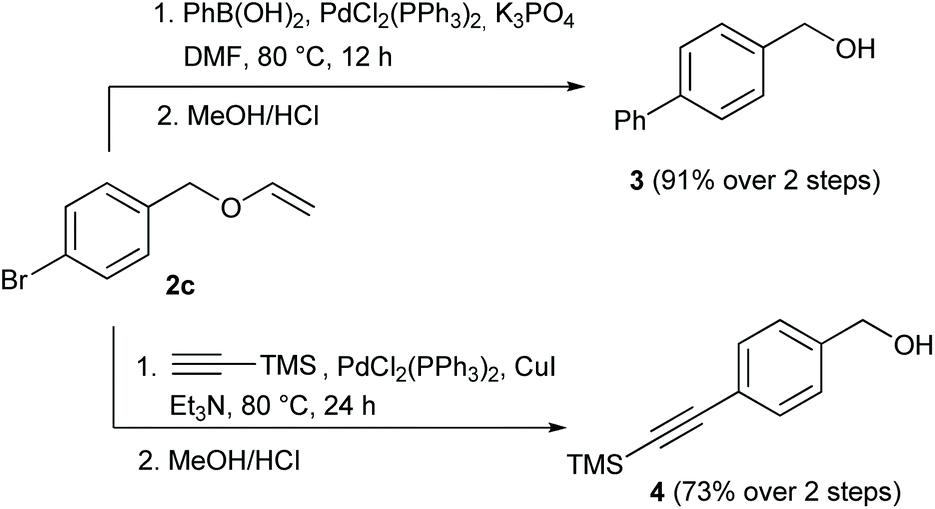
Examining the vinyl moiety as a protecting group for hydroxyl (–OH) functionality under basic conditions - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D0QO00202J
![Poly[di(ethylene glycol) vinyl ether]-stabilized poly(vinyl acetate) nanoparticles with various morphologies via RAFT aqueous emulsion polymerization of vinyl acetate | Polymer Journal Poly[di(ethylene glycol) vinyl ether]-stabilized poly(vinyl acetate) nanoparticles with various morphologies via RAFT aqueous emulsion polymerization of vinyl acetate | Polymer Journal](https://media.springernature.com/full/springer-static/image/art%3A10.1038%2Fs41428-020-00417-3/MediaObjects/41428_2020_417_Figa_HTML.png)
Poly[di(ethylene glycol) vinyl ether]-stabilized poly(vinyl acetate) nanoparticles with various morphologies via RAFT aqueous emulsion polymerization of vinyl acetate | Polymer Journal

Controlled Radical Homopolymerization of Representative Cationically Polymerizable Vinyl Ethers | Journal of the American Chemical Society

Novel Peptidomimetics Containing a Vinyl Ester Moiety as Highly Potent and Selective Falcipain-2 Inhibitors | Journal of Medicinal Chemistry
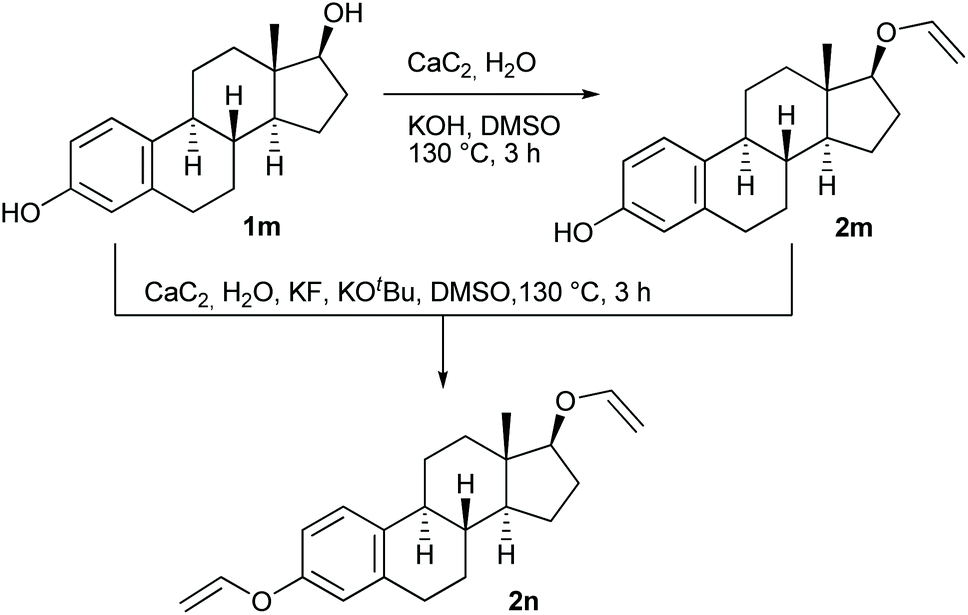
Examining the vinyl moiety as a protecting group for hydroxyl (–OH) functionality under basic conditions - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D0QO00202J

Rhodium-catalyzed mild and selective C–H allylation of indolines and indoles with 4-vinyl-1,3-dioxolan-2-one: facile access to indolic scaffolds with an allylic alcohol moiety - ScienceDirect

Wet carbonate-promoted radical arylation of vinyl pinacolboronates with diaryliodonium salts yields substituted olefins | Communications Chemistry
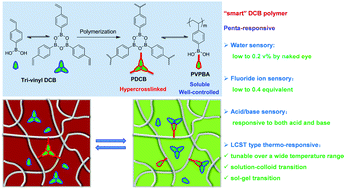
Well-controlled polymerization of tri-vinyl dynamic covalent boroxine monomer: one dynamic covalent boroxine moiety toward a tunable penta-responsive polymer - Polymer Chemistry (RSC Publishing)
![PDF] Vinylation of a Secondary Amine Core with Calcium Carbide for Efficient Post-Modification and Access to Polymeric Materials | Semantic Scholar PDF] Vinylation of a Secondary Amine Core with Calcium Carbide for Efficient Post-Modification and Access to Polymeric Materials | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f286ceb376de35cacaa09ad45f90c27e91883fe4/3-Table1-1.png)
PDF] Vinylation of a Secondary Amine Core with Calcium Carbide for Efficient Post-Modification and Access to Polymeric Materials | Semantic Scholar

Synthesis and structural characterization of new spiropyran containing conjugated vinyl-3Н-indolium moiety and its hydrolysis product | SpringerLink

Structure-activity relationships of cephalosporins having a (dimethylisoxazolidinio)vinyl moiety at their 3-position. | Semantic Scholar

The Vinyl Moiety as a Handle for Regiocontrol in the Preparation of Unsymmetrical 2,3‐Aliphatic‐Substituted Indoles and Pyrroles - Huestis - 2011 - Angewandte Chemie - Wiley Online Library

Radical polymerization behavior and thermal properties of vinyl ethylene carbonate derivatives bearing aromatic moieties - ScienceDirect

Microwave-Assisted Palladium-Catalyzed Cross-Coupling of Aryl and Vinyl Halides with H-Phosphonate Diesters

Examining the vinyl moiety as a protecting group for hydroxyl (–OH) functionality under basic conditions - ScienceDirect