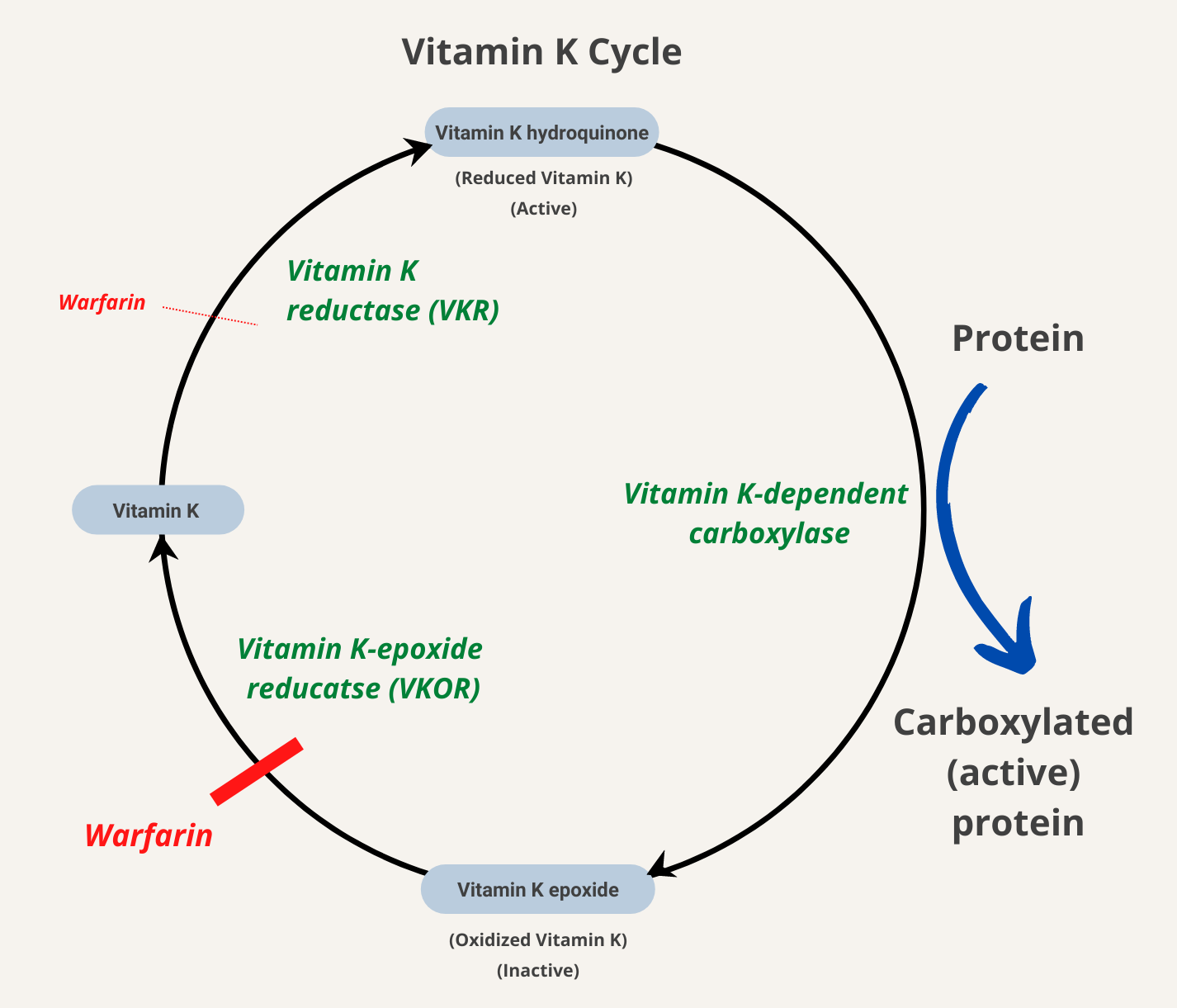
The vitamin K cycle. Abbreviations were used to define enzymes involved... | Download Scientific Diagram
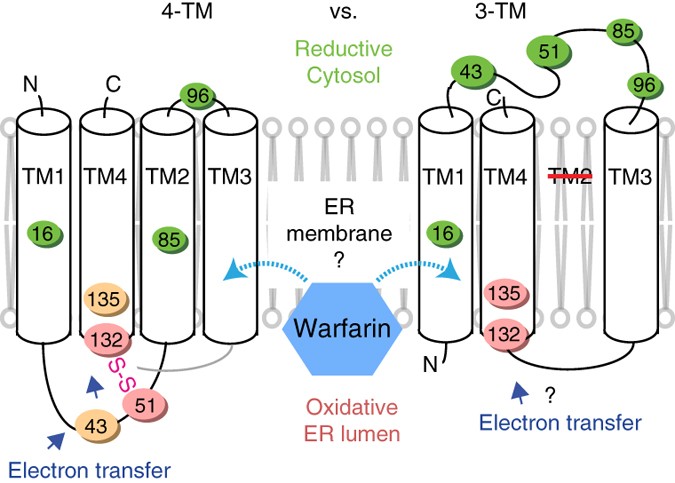
Structures of an intramembrane vitamin K epoxide reductase homolog reveal control mechanisms for electron transfer | Nature Communications
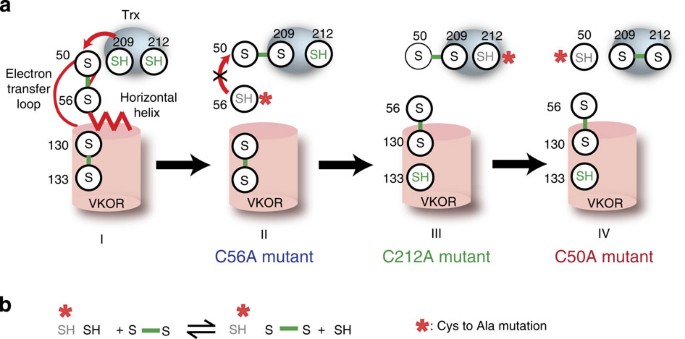
Warfarin traps human vitamin K epoxide reductase in an intermediate state during electron transfer | Nature Structural & Molecular Biology

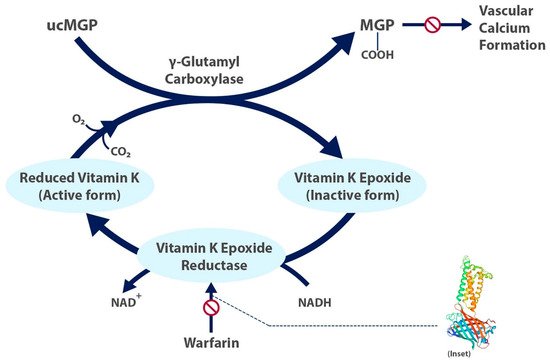
Vitamin K Epoxide Reductase Complex Subunit 1 (VKORC1) Polymorphism and Aortic Calcification | Arteriosclerosis, Thrombosis, and Vascular Biology

Figure 1 from Pesticide resistance-Mechanisms of anticoagulantin wild mammals resistance in wild rodents | Semantic Scholar
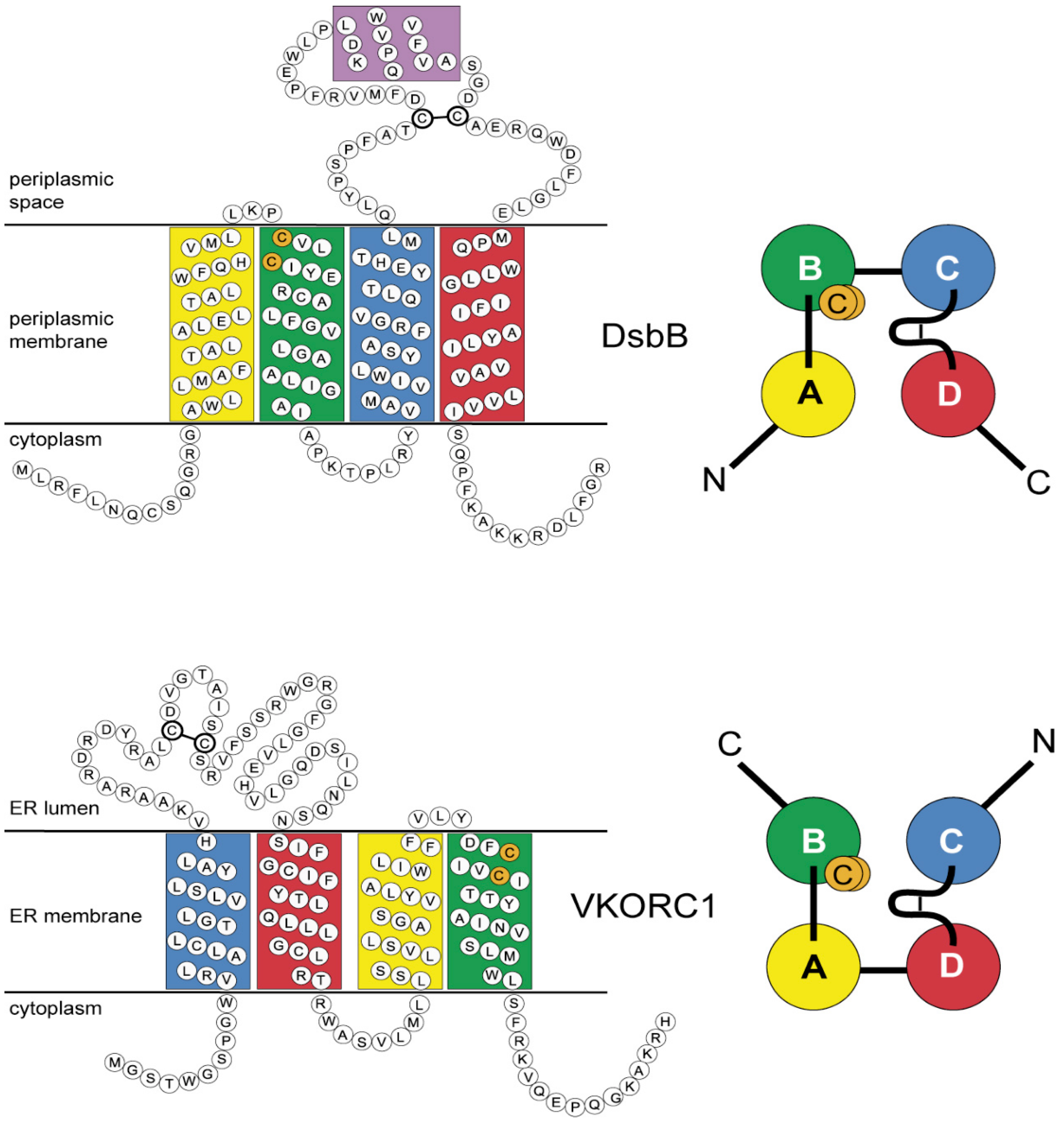
Nutrients | Free Full-Text | Phylogeny of the Vitamin K 2,3-Epoxide Reductase (VKOR) Family and Evolutionary Relationship to the Disulfide Bond Formation Protein B (DsbB) Family

Vitamin K Epoxide Reductase Complex Subunit 1 (VKORC1) Polymorphism and Aortic Calcification | Arteriosclerosis, Thrombosis, and Vascular Biology